

But there’s one drawback with this accessible jewelry - it leaves greenish-blue marks on the skin. This occurs because the metal is very quick to oxidize. Oxidation is the process where a material - in this case, copper - changes when it comes in contact with oxygen molecules in air or water. Oxidation is why many metal statues containing copper often get covered in a green patina over time. A widely known example is the Statue of Liberty.
In the case of your jewelry, however, the culprit behind the green stains is usually sweat, soap, or lotion. These liquids react with the jewelry and make it oxidize, turning the metal green. This is also why you will get those green stains on some days but not on others.
Interestingly, even jewelry that claims to be gold or silver sometimes tarnishes. That’s because very few jewelry pieces are actually made of 100% gold or silver; other materials, such as copper or nickel are usually mixed in as well for durability or price reduction.

First things first - if you get the green stains, don’t worry. They’re not dangerous and can be easily washed away with water. But if you’re looking to prevent the stains from recurring altogether, here are a few tips:
1. Avoid using lotion or soap when you have the jewelry on. For example, if you have to wash your hands, take off the ring before washing your hands and put it back on once your hands are clean and dry.
2. Don’t exercise while wearing copper jewelry or wear it in hot weather. Sweat can trigger copper oxidation, so avoid wearing your copper necklaces during a workout, a hike, or a swim.
3. Opt for metals that don’t oxidize, such as stainless steel, rhodium-plated jewelry, or silver and gold.
4. Coat the inside of the jewelry with clear nail polish. The coat of nail polish will protect the metal from oxidizing. This trick works wonders with copper rings, but it might be trickier to do with necklaces or jewelry items with intricate details. You can reapply when the initial coat of nail polish flakes off.
We hope you found this post useful.
If so, please share it with others!
H/T: Insider, Mental Floss

Can Wearing a Mask Protect You From the Coronavirus?
With each new virus outbreak, people start wearing masks as protection from the disease, but are these as effective as we think they are?

The Relationship Between Migraines and Weather Explained
Various weather conditions may trigger migraines in some people. Learn what weather triggers of migraines exist and how to always be prepared

The Royal Jewelry Collection: Let's Take a Peek...
The British royal family has some of the most spectacular pieces of jewelry in the world. Take a look at this post to see amazing tiaras, necklaces and earrings
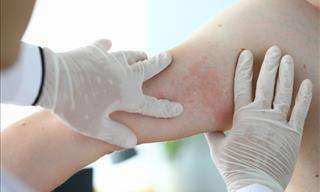
A Stress Rash Is a Real Thing, Here’s Why It’s Important!
It’s true, a stressful day can truly make some people break out in hives!
 6:29
6:29
4 Beginners Exercises to Improve Your Balance in 5 Minutes
This video tutorial will help you maintain a sense of balance and strength in just 5 minutes a day

4 Simple Ways to Open a Jar Lid That’s Stuck
Opening a jar can be quite tricky and sometimes seemingly impossible, really, but with these tricks, you'll be able to twist it open in no time!
 15:13
15:13
Cleaning Guide: The Correct Way to do Daily Chores
We could all do with a couple of life-savers every now and then. Check these out!

12 Fabulous Uses for Socks That Have Lost Their Mate
Here is a terrifically useful list of 12 handy ways that your odd sock or stocking can regain her purpose, get ‘out there’ and reinvent herself.
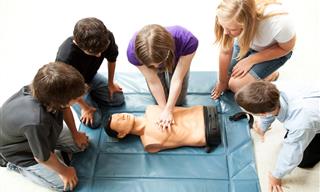
10 First-Aid Techniques Most of Us Perform Incorrectly
Performing first aid methods wrongly can do more damage than good. Here are 10 common first aid methods that are often performed incorrectly.

How to Clean Grout So It Looks Brand New
f your tile grout looks dingy, don't despair—you can quickly restore its appearance with proper cleaning methods.

Banish Yeast & Bad Bacteria with These 7 Natural Solutions
Yeast and bad bacteria can be very harmful as well as embarrassing, so here are 7 wonderfully natural preventative remedies.

Warning! Eating Too Much Licorice Can Damage Your Health!
Eating too much licorice can be dangerous. Read here to find out more.

How To Ward Off the Most Common Summer Pest
In order to prevent unwanted pests in summer - especially cockroaches - consider this one important thing.
 14:45
14:45
These Tricks will Transform Your Trash into Treasure!
This fun new method of reusing plastic bottles helps you make them into beautiful homes for all your plants

40 Common Tourist Traps to Avoid on Your Next Vacation!
If you're planning a trip abroad, please be aware of these 40 common tourist scams!

Read This to Deal With That Terrible Smell In Your Fridge!
If there's a bad odor in your refrigerator, then this guide will show you exactly how to get rid of it quickly and easily.

Why You Shouldn’t Keep a Bar Soap for More Than 2 Years
The following 10 personal hygiene products all expire sooner than you’d think and you must replace them regularly
 3:15
3:15
Cleaning Tips: The Best Ways to Save Your Burnt Pot
Cleaning a scorched pot without scrubbing is possible! Here are some tips to make it look as good as new with ease.

Great Dog Training Tips You'll Wish You Knew Before!
Here are 10 great training secrets that will make it easier for you to train your puppy. Take a look.

20 Cooking Tips Everyone Should Be Aware Of
These cooking tips will hopefully close a lot of little holes in your kitchen knowledge.
 13:03
13:03
Running Out of Storage Space? Keep These 20 Tips in Mind
Running out of storage ideas? Here are 20 space saving ideas to bear in mind.

Wow! Even We Were Amazed At These 11 Ingenious Tips
Everyone needs some tips in life, and these 11 are worth remembering because they'll help you solve a variety of problems, make life easier and save money!

Plagued By Liver Spots? Here Are 9 Ways to Get Rid of Them
Skin spots are a reality for many of us and can often be a nuisance. But, fortunately, they can be lightened with these 9 natural ingredients.

If You Value Your Health, Wash These 9 Foods Well
Are you washing these 9 foods correctly? Probably not!
 0:48
0:48
One Minute DIY: Turn Old Jeans Into a Useful Garden Apron
If you don't like wasting money or old clothes this video is for you! Learn in 1 minute how to transform your jeans into a terrific garden apron.

Learn About 12 Popular Spices in Their Natural Form
We've gathered 12 very common spices and included photos showing them as plants blooming in nature.
 7:09
7:09
How to Slice Cooked Meat For the Tenderest Results!
Cutting cooked meat properly is just as important as cooking it right. Find out how to slice beef, pork, and poultry for the tenderest results!
 8:55
8:55
21 Awesome Kitchen Hacks That are Really Useful
From using a lighter to open up a stiff jar to learning how to sharpen a knife in no time at all, these life hacks will make your life easier.

We All Have Aspirin - And Here Are 9 Things to Use It For
Aspirin is about as pedestrian a pill as you can think of - or so you might think! In reality it has loads of surprising uses. Here are 9 of the best.

These Flowers Add Color With Very Little Gardening Effort
If you want to have a colorful summer garden but don't fancy dedicating all your free time to it, all you need are these 12 flower varieties. Read on for more.

8 Shopping Tricks That Will Save You Money
Learn to save more money with these secret tips.

From Leather to Linen - How to Clean Every Type of Couch
Couches tend to collect dirt, but cleaning them can be tricky. Take a look at these expert-approved tips on how to clean 8 different upholstery materials.

7 Scented Flowers for a Sweet Smelling Home
Opt for these beautifully scented flowers as opposed to air fresheners or candles for a better smelling home.

Home Hacks: 8 Smart Tips to Declutter Your Bedroom
Take note of these handy tips to achieve an organized and clutter-free bedroom.
 10:02
10:02
5 Antiques That Are NO Longer Worth the Investment
People collect various antiques, hoping that their collection of dolls, furniture, or cards is worth a pretty penny... Watch this before you decide to sell them.

These Surprising Garlic Uses Show It's More Than Just Food
While garlic is an important ally for natural health, it has many more functions. Check out these 14 unusual alternative uses for garlic.

Making Laundry Products At Home Is EASY - Here Are 8 Ideas
In this article, we show you how to make homemade laundry detergent, laundry pods, and dryer balls. We also share many more useful laundry tricks.

Teach Your Dog 10 Basic and Important Commands!
There are numerous ways you can teach your dog tricks and commands, however the best way is to use a clicker. Learn how to teach your dog new tricks.
 13:42
13:42
Flying Soon? These Clothing Choices Are a Bad Idea
This guide covers what not to wear when going to an airport.
 10:19
10:19
These Simple Life Hacks Will Make Your Life So Much Easier
These 19 simple life hacks are bound to make your life a lot easier. Take a look and give them a go yourself!

The Labels on a Plastic Bottle Are There for Your Own Good
Plastic bottles of them have symbols on them for a reason, and these reasons are in order to ensure your health. Heare is a complete guide.

You MUST Pay Attention to These Numbers When Buying Fruit
The numbers you see on fruit have much more significance than you think. You can learn everything you need to know about the fruit you're buying from them.

Organizing Mistakes That Make Your Kitchen Look Cluttered
Take a look at the possible organizational mistakes you are making that are making your kitchen look messy.

Cinnamon Isn't Just Delicious, It's Also Incredibly Helpful!
The best uses for cinnamon aren't necessarily in food, many of its benefits derive from the spice's anti-fungal, antibacterial and antioxidant prowess.
 5:18
5:18
Make Artificial Snow From a Nappy Diaper!
We're going to teach you a super easy way to make fake snow this holiday, and all you need are some diapers,

The Ultimate Guide to Stress-Free Family Holiday Travel
Enjoy holiday travel with your family this season without the usual headaches.

How to Care for Indoor Plants During the Winter - 6 Tips
Caring for your houseplants in the winter months can be quite tricky. Here are some useful tips that can help.
To enable your Ad-Free Subscription, please fill the fields below
Your subscription was successful, now you can enjoy an ad-free experience!! Note: To make sure you get no ads, please make sure to log in to your account. If you are logged in already, then refresh the page. The subscription can be cancelled at any time.


