
If you use over-the-counter eye drops, this is important news. A major recall is currently underway, and the products in question have been sold at some of the most popular pharmacies in the country.
A California-based pharmaceutical company, K.C. Pharmaceuticals, has voluntarily recalled more than 3.1 million bottles of over-the-counter eye drops. The recall was initiated in early March and formally classified by the Food and Drug Administration (FDA) shortly after.
The reason? Concerns about sterility — specifically, a potential lapse in the manufacturing process that could compromise the cleanliness of the products. Importantly, no illnesses or injuries have been reported so far, but experts say caution is absolutely warranted.
Which Products Are Affected?
The recalled eye drops were sold at major retailers including Walgreens, Kroger, and CVS. If you have any of the following products at home, you should stop using them immediately and throw them away:
CVS has confirmed that customers who purchased any of the four recalled items sold through their stores may return them for a full refund.
How Serious Is This?
The FDA classified this as a "Class II" recall, which means that while using the product could cause temporary health consequences, the likelihood of a serious health problem is considered remote. Think of it as a precautionary measure rather than a confirmed danger.
That said, the word "sterile" on a label matters a great deal when it comes to eye drops. Unlike other body parts, your eyes are extremely vulnerable to infection, and any product that goes into them must be completely free of bacteria and other microbes. As one expert put it, "We don't know if they really were contaminated. If they were, we don't know what type of microbe. All we know is that the manufacturer said there's an issue with sterility in these products."
A Reminder of Why Eye Drop Safety Matters

This recall brings back troubling memories of a serious outbreak just a few years ago, in 2023, when contaminated over-the-counter eye drops were linked to more than 80 infections across the United States, 14 cases of permanent vision loss, and four deaths. The bacteria responsible — Pseudomonas aeruginosa — was antibiotic-resistant, making it especially dangerous.
That incident was a wake-up call about the importance of manufacturing standards for eye care products — and a reminder that "over-the-counter" doesn't always mean risk-free.
What About Prescription Eye Drops?
If you use prescription eye drops — for glaucoma, dry eye, or an eye infection — you have nothing to worry about. This recall applies only to over-the-counter products. Doctors are urging patients to continue using their prescription medications without interruption.
Signs of an Eye Infection to Watch For
If you've recently used any of the recalled products, keep an eye out (pun intended) for these warning signs:
Keep in mind that these symptoms can sometimes be caused by seasonal allergies rather than an infection, especially at this time of year. The key is to monitor how things progress — if symptoms don't improve, it's time to see a doctor. And if you wear contact lenses, be especially vigilant. Contact lens wearers face a higher risk of eye infections because tiny, unnoticed abrasions on the eye can make them more susceptible to bacteria.
Simple Habits That Protect Your Eyes
Whether or not your eye drops are part of this recall, these best practices can help keep your eyes safe every time you use them:
The bottom line: toss any affected products, watch for symptoms, and don't hesitate to contact your doctor or pharmacist if you have any concerns. Your vision is too precious to take chances with.

7 Happy News Stories You Won't Be Able to Resist Sharing
Watching the news lately can make you feel like only terrible things are happening in the world, so here are 7 really good things that are going on right now.

Do Hangovers Really Get Worse With Age?
While research on this phenomenon is somewhat limited, science offers several compelling explanations for why that morning-after misery intensifies as the years go by.

Country Fresh Fruit Packets at Walmart Recalled By the FDA
Equipment found contaminated with bacteria has prompted a voluntary recall of Country Fresh fruit products from Walmart outlets.

Why Are Cancer Patients Less at Risk of Alzheimer's?
Doctors and researchers began noticing something strange: people who had been diagnosed with cancer seemed less likely to develop Alzheimer's later in life. And conversely, Alzheimer's patients seemed to have lower rates of cancer.

Avoid These Ingredients If You Love Your Mental Health...
Here are some of the worst kinds of food that you can eat if you care about your cognitive health.

Can We PREVENT Type 1 Diabetes?
The study's findings point toward developing a simple blood test that could detect the immune failure leading to type 1 diabetes before insulin-producing cells are permanently lost.
 4:16
4:16
Feeling Bloated? This Is How to Improve Digestion Quickly
How can you improve your digestion quickly? This guide explains.
 5:41
5:41
Suffer From Joint Pain? You Should Eat More Apple Peels
Could eating more apple peels reduce the effects of chronic joint pain? This video explains.
 10:21
10:21
See How Germs Spread With Your Own Eyes
If you need to see it to believe it - this visual demonstration of how germs spread is for you!

Basil Seeds vs. Chia Seeds: A Nutritional Comparison
While chia seeds have long been celebrated as a nutritional powerhouse, basil seeds are emerging as a worthy competitor. Both offer impressive health benefits, but which one deserves a spot in your pantry?

GUIDE: Is GLP-1 Right For You?
Glucagon-like peptide-1 (GLP-1) medications, which mimic a natural gut hormone that helps regulate appetite and blood sugar, are increasingly appearing in discussions about weight, metabolism, and long-term health.
 5:05
5:05
Frustrated With Your Dandruff? Here’s How to Get Rid Of it
Dandruff can be quite irritating, particularly in the winter months. Learn what causes it and you can get rid of it.

Which Drinks Increase Your Risk of Stroke?
Let’s see what exactly these studies say, which drinks are worth drinking more of, and which ones are best to avoid.
 3:17
3:17
Brilliant Doctor Easily Fixes This Common Injury!
Learn all about plantar fasciitis, a common foot injury, and discover the best non-surgical way to treat it.

The Health Benefits of Garlic and Honey: Nature's Powerful Duo
Garlic and honey are two of nature's most treasured foods, used for centuries to support health and wellness.
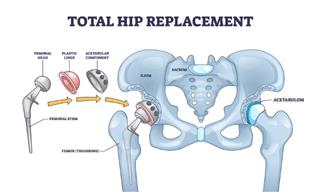
Medical Guide: All About Hip Replacement Surgery
Hip replacement surgery can alleviate pain, restore hip function, and improve mobility and quality of life.
 7:10
7:10
Exercise Your Knees at Home to Prevent Recurring Pain
Knee pain is one of the most common pains, but these simple exercises can help you out...

If You Eat These 9 Fish Regularly, You Should Stop!
We are always told how fish is healthy, but there are actually some types of fish that'll do more harm than good. Here are nine of them!
 5:01
5:01
How to Balance the Damage from Eating Refined Carbs
Refined carbs can cause some serious health effects. This is what you should do to balance the damage.
 14:14
14:14
Expert Nutritionist Tackles Your Most Burning Diet Q's
Nutritionist Dr. David Katz is here to answer real diet questions from the internet.

Can a 'Cortisol Cocktail' Actually Reduce Stress?
This trendy beverage, dubbed the "cortisol cocktail," has taken the wellness world by storm.

Diabetics at a Holiday Party: Essential Tips
Holiday parties don't have to derail your diabetes management. With smart planning and mindful choices, you can enjoy celebrations while keeping your blood sugar stable.

How 25% of Patients Managed to Prevent Diabetes
How long does it take to get rid of prediabetes, and how is it done? That's exactly what we'll answer in the following article.
 12:29
12:29
Health Problems Your Feet Might Be Telling You About
Pay close attention to your feet. They might be telling you about some hidden health issues.

Hidden Danger: Understanding and Combating Visceral Fat
Visceral fat, the dangerous adipose tissue that accumulates around vital organs like the heart, liver, and kidneys, represents one of the most significant health risks of our time.

The Full List of Herbs That Protect You From Diabetes
Diabetes is dangerous, therefore it's important to be mindful of our food intake and include these 15 diabetic herbs and spices in our diet!

Understanding the Early Detection of Breast Cancer
When breast cancer is discovered in its early stages, treatment success rates are remarkably high.

8 International Health Paradoxes that Got Me Thinking...
Here are some of the most notable and interesting food paradoxes that will get you thinking about the way you eat (and possibly where you live)!

Castor Oil For Hair Loss: the Facts
Is there truth to the claims about castor oil fighting hair loss? Let's find out

A Simple Sleep Habit That Could Lower Your Blood Pressure
New research suggests that going to bed at the same time every night may help reduce blood pressure and improve heart health
 14:07
14:07
Feeling Anxious During Bedtime? This Guide Will Help
Felling anxious while going to bed? Listen to the tips shared by this therapist on how to train your brain to worry less.

Can We PREVENT Type 1 Diabetes?
The study's findings point toward developing a simple blood test that could detect the immune failure leading to type 1 diabetes before insulin-producing cells are permanently lost.
9 Habits That Protect You From Fungal Infections
If you suffer from some of these symptoms, there are a number of home remedies that can easily treat fungal infections.

Sleeping Risk: How to Save Your Brain While At Rest
For the first time, researchers have identified the effect of our breathing rate during sleep.

Can What You Eat Raise Our Risk of Parkinson’s?
An extensive American study has found that daily consumption of ultra-processed foods—such as snacks, hot dogs, sugary drinks, and packaged pastries—may double the risk of developing early neurological symptoms linked to Parkinson’s disease.

How Ear Reflexology Can Help Relieve Aches and Pains
If you suffer from aches and pains in any part of your body, then why not give ear reflexology a try?

MDMA and Magic Mushrooms: New Hope for PTSD Treatment
In recent years, scientists have turned to an unlikely source for a breakthrough in PTSD: psychedelic drugs once known mainly for recreational use.
 3:33
3:33
What Causes Constipation? Find Out Here...
What's behind constipation? This informative TED-Ed video sheds some light on the matter!
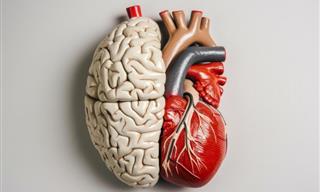
Heart Damage in Midlife May Increase Risk of Dementia
A new study reveals that a blood test typically used to detect heart attacks could help identify who might develop dementia decades in the future.

9 Effective Treatments for Stiff and Sore Muscles
Have you ever woken up in the morning and stretched only to feel neck or shoulder pain? here's how you treat and prevent it.
 5:11
5:11
Blueberries: Nourishing Your Body the Natural Way
Blueberries are nutritious, anti-inflammatory and rich in antioxidants, a true dietary medicine.
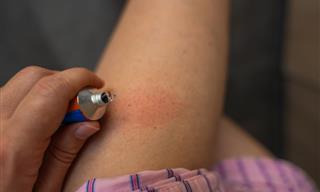
How to Prevent Painful Chafing: Five Natural Solutions
It’s even possible to prevent chafing in advance if you know what to do and how to prepare for it. We will teach you all about it here – the signs, home treatments, and prevention methods.

10 of Grandma’s Remedies for Mature and Loose Skin
Although most of us experience changes in our skin's appearance, some of them can be prevented and improved with The following 10 grandma's remedies!

What to Know About Acupuncture for High Blood Pressure
Acupuncture, a healing technique rooted in traditional Chinese medicine (TCM), may help temporarily lower blood pressure by reducing stress and influencing your body's hormones.
 23:23
23:23
How Food Can Prevent Alzheimer’s and Boost Brain Health
Let’s uncover the power of food to transform your mind and well-being.
 5:21
5:21
A Few Facts You Probably Don’t Know On Salmon
Salmon - the different varieties, health benefits and more interesting facts you probably didn’t know about this popular fish
 7:25
7:25
What REALLY Happens When We Go Under Anesthesia
Learn how anesthetic drugs work on your brain, why you don't remember anything, and how it differs from natural sleep.
To enable your Ad-Free Subscription, please fill the fields below
Your subscription was successful, now you can enjoy an ad-free experience!! Note: To make sure you get no ads, please make sure to log in to your account. If you are logged in already, then refresh the page. The subscription can be cancelled at any time.


