How is type 1 diabetes unique?
Diabetes is an umbrella term for three separate conditions:
- Type 1 diabetes - a hereditary autoimmune condition
- Type 2 diabetes - the most widespread, may be acquired or inherited
- Gestational diabetes - diabetes that appears in pregnancy.
Of these three conditions, type 1 diabetes is the most difficult to treat. While gestational and type 2 diabetes can often be managed with changes in diet, exercise, and a variety of medications, the only available treatment option for type 1 diabetes is insulin.
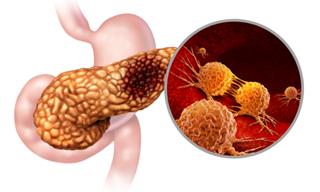
In type 1 diabetes, the immune system attacks and destroys the cells in the pancreas responsible for producing insulin called beta cells. As a result, the pancreas is able to produce less and less insulin, which makes blood glucose levels rise. Insulin is a hormone produced in the pancreas that allows glucose to enter the cells and be utilized as energy. When the levels of insulin drop, glucose levels skyrocket and eventually damage the body.
Type 1 diabetes is usually diagnosed in children and young adults. A person is at higher risk for type 1 diabetes if they have a relative with type 1 diabetes. This is precisely the group of patients Teplizumab-mzwv can benefit.
Related Article: Type 1 & Type 2 Diabetes - What Are the Stages & Symptoms?
How does Teplizumab-mzwv work?
Teplizumab-mzwv is the first medication capable of delaying the progression of type 1 diabetes. The treatment was approved by the US Food and Drug Administration (FDA) on November 17, 2022, under the brand name Tzield.
Tzield is a synthetic protein that works like a monoclonal antibody produced by the immune system. This is a type of antibody that instructs immune T cells to ignore the beta cells of the pancreas - states ProventionBio, the manufacturer of Teplizumab-mzwv. In addition, the medication urges the immune system to produce more immune cells that protect the beta cells from T cells.
Who can benefit from Teplizumab-mzwv?
The treatment is designed to slow down the progression of the disease. Therefore, it is beneficial for patients prior to the 3rd stage. Type 1 diabetes is formally divided into three stages that reflect the extent of damage to the beta cells and symptoms.
- Stage 1 is asymptomatic. It is characterized by elevated levels of antibodies that attack the pancreas, but blood sugar levels remain normal. Most often, it is diagnosed in children whose parents suffer from diabetes, and it can last for years.

- Stage 2 is also asymptomatic, but the patient’s glucose levels start to rise, suggesting a higher degree of beta cell damage. Stage 2 can last from months to years.
- Stage 3 is the last. Patients notice symptoms like blurry vision, increased thirst and urination, fatigue, and weight loss. The pancreas can no longer produce enough insulin to sustain the body, and the patient requires supplemental insulin.
Teplizumab-mzwv is approved for use in all patients with stage 2 type 1 diabetes who are eight or more years old. The medication was shown to delay the advance to stage 3 for more than two years compared to a placebo in a clinical trial. There was some variability among participants, meaning some patients developed stage 3 later and others sooner.
"The drug's potential to delay clinical diagnosis of type 1 diabetes may provide patients with months to years without the burdens of disease," said Dr. John Sharretts of the FDA Center for Drug Evaluation and Research in an official press release of the FDA.
The Medical Trial

Seventy-six participants aged 8-49 years old were involved in the trial. All had stage 2 type 1 diabetes. Forty-four of the participants received IV infusions of teplizumab-mzwv for 14 consecutive days, and the remaining participants got placebo infusions.
After four years and three months, 45% of patients who received teplizumab-mzwv were diagnosed with stage 3 compared to 72% of the placebo group. On average, placebo patients developed stage 3 type 1 diabetes twice as fast as those who got the treatment.
Common side effects of teplizumab-mzwv
A rash and headaches are the most common side effects of teplizumab-mzwv. Patients who undergo the immunotherapy may be more susceptible to infections because teplizumab-mzwv can temporarily lower the body’s immune protection.
Some patients in the trial developed a temporary decrease in some immune cells. According to ProventionBio, the number of these immune cells started recovering a week after the treatment and fully bounced back a couple of months later.

The manufacturer also cautions that patients who take teplizumab-mzwv should be monitored by a professional due to more serious potential side effects like vomiting or angioedema (swelling under the skin). Sometimes, teplizumab-mzwv triggers something called cytokine release syndrome - an acute syndrome in which T cells cause a systemic inflammatory response that comes with a fever and multiple organ dysfunction.
How much does teplizumab-mzwv cost?
The USA Today reports that a single vial of Teplizumab-mzwv costs $13,850, so the full 14-day treatment is worth $193,000. This is a steep price tag, but many people now have the potential to delay the disease and the burden it creates every day, and for them, teplizumab-mzwv is a life-changer.
 Go to BabaMail
Go to BabaMail