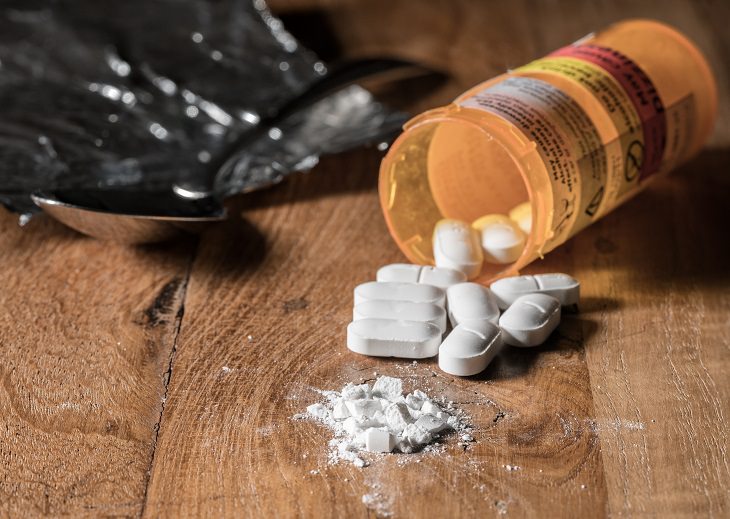
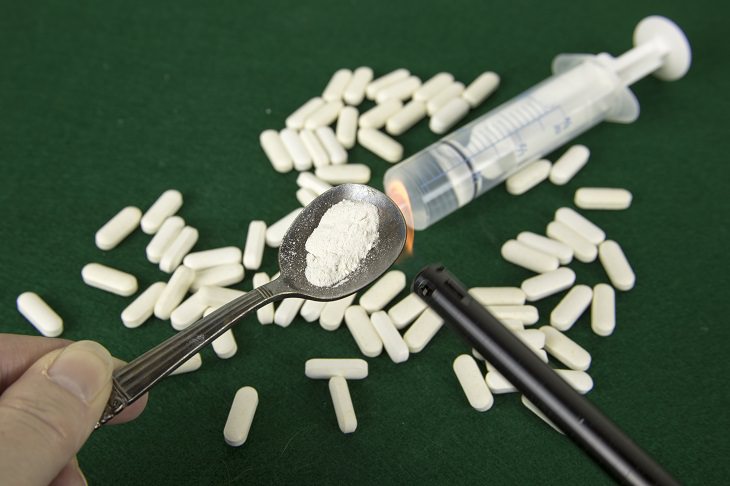
America is in the midst of the worst opioid epidemic in its history, and what’s most alarming is that the drugs most complicit in the plague of addiction and overdoses are perfectly legal and available in most pharmacies, including cough syrup, fentanyl, oxycodone, and others. Now, instead of holding back this epidemic, an opioid stronger than anything we’ve ever known may well be making its way to the market.
Dsuvia is a powerful painkiller that was approved by the FDA last year for use in supervised settings, such as ERs, specifically for use in cases of severe physical trauma, such as a broken bone or gunshot wound.
Just how strong is this stuff?
Dsuvia is about 10 times stronger than fentanyl and 1,000 times more powerful than morphine!
But hold on, if Dsuvia has been approved for hospital use only, then what’s the problem anyway?
The problem is twofold: drug diversion and misprescription, both potentially leading to lethal results.
Drug diversion is the umbrella term for illegal use of legally-prescribed medicine for any use that is not the one it was intended for. Obtaining such prescription drugs is achieved by criminals in several ways, including “doctor shopping” - the act of visiting a doctor with false health complaints and with the intent of receiving a specific opioid prescription, theft and forged prescriptions.
Misprescription is the act of prescribing either the wrong drug or the wrong dose. This can be especially disastrous in the case of opioids that are extremely potent and addictive. According to a recent study published in JAMA, between 35% and 55% of patients that are prescribed fentanyl are actually opioid intolerant. For reference, fentanyl accounts for about half of overdose-related deaths in the United States.
Because Dsuvia comes in small, soluble tablets, medical experts are worried it will be easier to divert and abuse. And there does not seem to be any serious medical need for it, either, as the same drug already exists in hospitals in injectable form.
And one cannot simply ignore the market forces at play in creating the current health crisis, as drug manufacturers have every commercial reason to encourage sales by decreasing costs, advertising the drugs and fiddling with prescription practices to make the drug easier to obtain. There is no reason to believe Dsuvia will be any different, as its distribution will be monitored by the manufacturer, AcelRX.
Dsuvia will be regulated in accordance with the FDA’s REMS (risk evaluation and management strategies) program for the monitoring of high-risk drugs, but before you take a sigh of relief, consider that the lethal fentanyl is also monitored by the same program, proving REM’s limited efficacy in stopping the abuse of dangerous opioids.
 Go to BabaMail
Go to BabaMail