Diabetes medications have gone a long way. Just imagine, before the invention of insulin in 1921, there wasn’t much doctors could do for diabetics. And now, a mere century later, most diabetes patients have a variety of treatments at their disposal and can safely manage their condition at home.
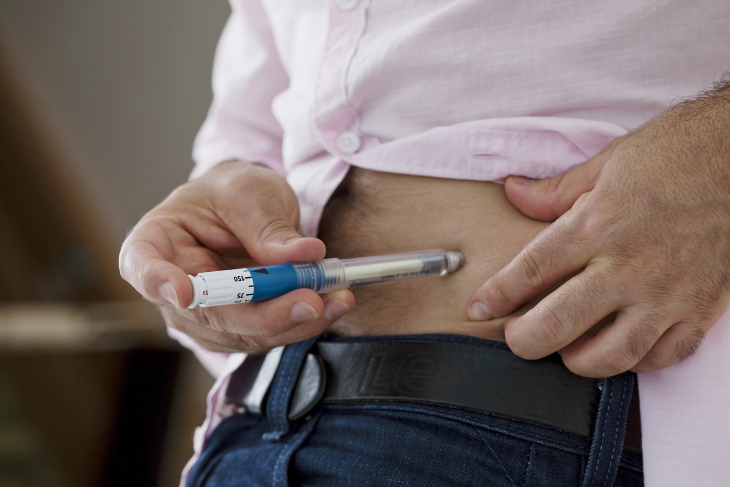
Still, patients who rely on insulin are significantly limited by their condition even today. Many require injecting insulin twice daily, which can be tricky to keep up with and may lead patients to miss a dose. But the solution may not be far away. The American Food and Drug Administration recently approved a medication called tirzepatide (Mounjaro) that may replace insulin for many type two diabetes patients.
How is tirzepatide different from diabetes medications available on the market?
Medical experts are excited about tirzepatide because it is an insulin alternative than can be administered at home only once a week. The medication may also benefit patients who need to lose weight due to the condition.
The market name of tirzepatide is Mounjaro, and it is produced by Eli Lilly, an American pharmaceutical company. Mounjaro is a hormonal therapy that regulates the two hormones that regulate blood sugar: glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1).
By targeting these hormones, tirzepatide can reduce glucose levels after meals and during fasting, and it can promote weight loss as well. In the clinical trials leading up to FDA approval, patients who took 15 mg of tirzepatide lost 12-25 pounds and saw a 1.6% decrease in HbA1c, a blood sugar level measure.
Some patients may also be indicated to combine tirzepatide with insulin. The clinical trials found that such patients had an average of 1.5% reduction in HbA1c compared with control subjects who took a combination of insulin and placebo.
How is this medication taken and who can take it?
The medication is administered through an auto-injector pen once a week, and it is available in six doses from 2.5 mg to 15 mg per dosage. The pen is easy to use, so patients will be able to keep up with their medication from home. Needless to say, this is a massive improvement for people who currently need to take insulin daily or twice a day.
The FDA points out that tirzepatide works best when combined with healthy lifestyle choices, such as a balanced diet and a steady exercise routine. The medication may cause digestive side effects, namely:
- Decreased appetite
- Nausea and vomiting
- Diarrhea
- Constipation
- Abdominal pain or discomfort.
Mounjaro is not recommended by the FDA for patients with a history of medullary thyroid cancer and multiple endocrine neoplasia syndrome (type 2). They also point out that the medication has contributed to thyroid C-cell tumors in animal models, and it is currently unknown if there is an increased risk in humans too.
When will Mounjaro come to the market?
The exact date isn’t known yet, but it should be around pretty soon given that it has already been approved by the FDA. The price of Mounjaro is another unknown - for now.
Experts point out that Mounjaro will likely be available for patients who have Medicare or Medicaid via a prior authorization. Ask your healthcare provider if you’re eligible to get this type of authorization.
H/T: Verywell Health, FDA
 Go to BabaMail
Go to BabaMail